Ammonia – Viking Pump TSM420: HL-LL 4925 User Manual
Page 16
SECTION TSM 420
ISSUE
E
PAGE 16 OF 18
TABLE 1
* (Number of Btu’s to change one pound liquid ammonia
from liquid to gas)
B. Setting too high - reduce setting or get lighter
spring
C. Valve poppet binding - remove poppet, check for
burrs or foreign material
D. Shut-off valve in relief valve return line closed open
valve (would apply only to pumps with return-to-tank
type relief valves)
Ammonia (Anhydrous Ammonia, NH3)
Ammonia is a colorless gas or liquid, has a pungent odor, as
a gas is lighter than air, is easily liquefied by pressure alone
and is very soluble in water or alcohol
Ammonia is one of the best known and widely used
refrigerants in use today in ice plants, food lockers, cold
storage warehouses and other industrial cooling processes
Ammonia has a higher refrigeration effect, per unit of liquid
volume, than any other type of commonly used refrigerant;
other advantages are low initial cost and low pipe friction
losses
Ammonia, like LP-Gas and other vapor high pressure liquids,
has to be kept in a closed container to keep it from boiling
away For a given temperature the pressure built up within
the container is equal to the vapor pressure or saturation
pressure Conversely for any given pressure there is a
temperature at which the liquid and vapor are in equilibrium
(saturation temperature);
see table 1. If the pressure in
the container is held constant and the vapor is withdrawn,
the ammonia will vaporize (boil) As it boils it picks up heat
from the area around the container since over 500 Btu’s
are necessary to change one pound of ammonia from liquid
to vapor If the pressure over the ammonia is reduced, the
boiling temperature will be reduced, the boiling temperature
will be lowered
grades of Ammonia Available - commercial grade 99 5%,
refrigeration grade 99 95%
Explosive Limit in Air, by Volume - 16 0 to 25%
Corrosion - Ammonia will not attack iron or steel ever in the
presence of moisture; it will attack copper, brass, bronze and
zinc in the presence of moisture
Toxicity - Concentrations of ammonia in air as low as 53
parts per million can be detected by the sharp penetrating
odor
Exposure to ammonia causes intense irritation to the surface
tissues of the eyes, nose, throat and lungs Exposure to high
concentration of ammonia may blind, burn, strangle or kill
The effect of ammonia on the skin is that of a caustic burn,
varying in severity with the concentration of ammonia and
the length of time exposed Changes in respiratory and heart
action produced as reflex actions resulting from the irritation
of the respiratory tract
For information on the safe handling of ammonia,
see SAfE
PRACTICES, page 5.
AMMONIA
PHYSICAL PROPERTIES
Boiling point (atmospheric pressure)
Freezing point (atmospheric pressure)
Viscosity at -28°F
Specified Gravity at -28°F
Liquid Density at -28°F
Liquid Density at -28°F
* Latent heat of vaporization at -28°F
-28°F
-107 9°F
0 27 centipose
0 68
42 6 lbs /gal
5 7 lbs /gal
589 BTU
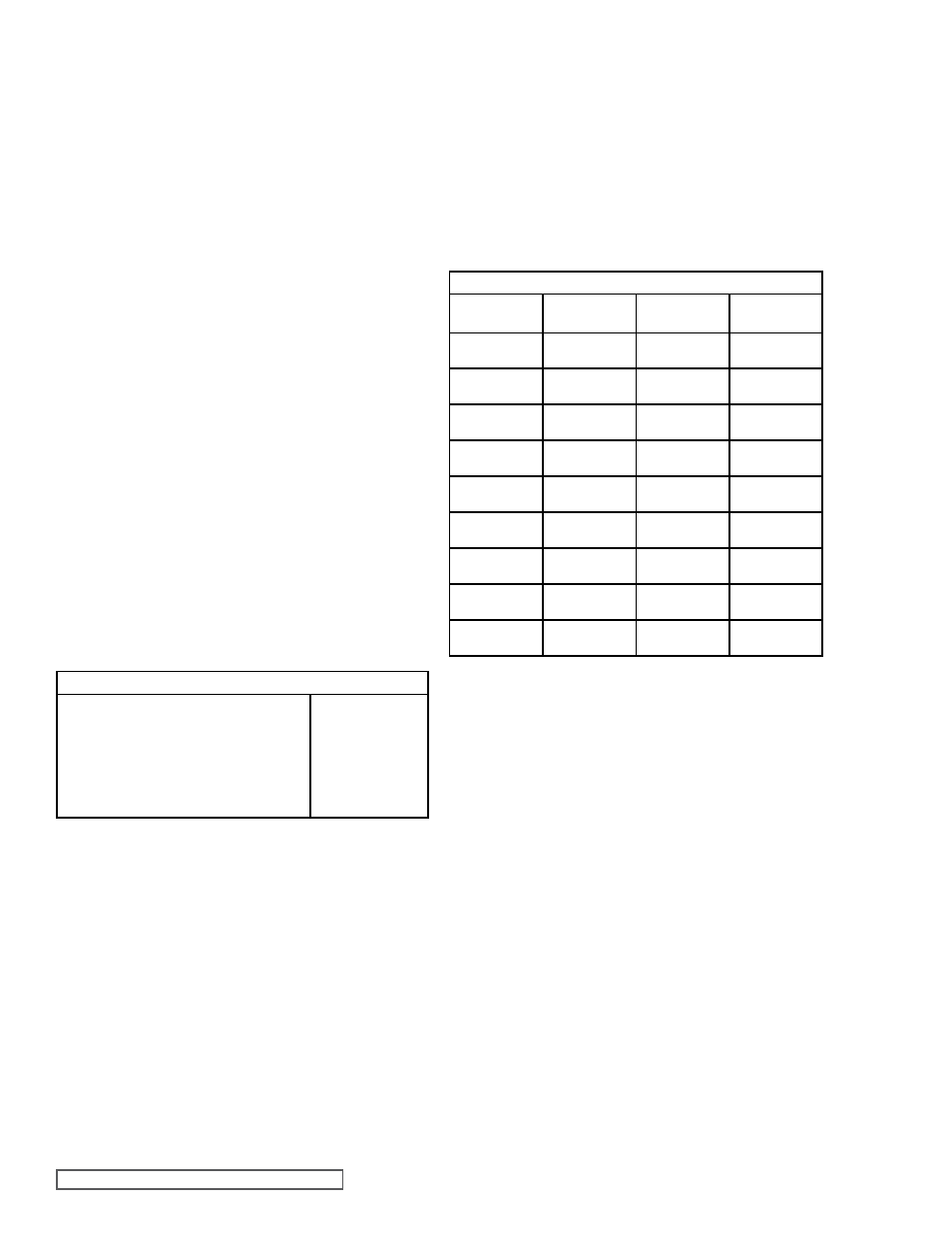
SATURATED VAPOR PRESSURE
Temp
°F
“Hg
Vacuum
PSI
Gage
PSI
Absolute
-100
180
27 4
24 3
1 24
2 74
-60
-50
18 6
14 3
5 55
7 67
-40
-30
8 7
1 6
10 4
13 9
-28
-20
0
0
3 6
14 7
18 3
-10
0
9 0
15 7
23 7
30 4
10
20
23 8
33 5
38 5
48 2
30
40
45 0
58 6
59 7
73 3
50
60
74 5
92 9
89 2
108
80
100
138
197
153
212
7. MECHANICAL SEAL LEAK.
A. Normal wear - replace seal
B. Improper installation - review installation procedure
outlined on pages 11 and 12
C. Abrasive material in liquid - if abrasive material on
ammonia side, use traps or strainer to clean system;
if on oil side, change oil more frequently and use
clean oil
D. Pump operating at very low temperature causing oil
to become heavy - use lighter oil, use immersion
heater to heat oil or consider a packed pump
Thus the temperature around the container can be controlled
by the pressure maintained in the container
Table 2 shows
the ammonia vapor pressure for various temperatures
* The same phenomena takes place to lower the boiling point
of water when the pressure is lowered This explains the low
boiling point of water on a mountain top
The saturation properties of ammonia make it desirable for
use as a refrigerant The temperatures necessary for freezing
and keeping food products can be quiet easily achieved
with pressures or vacuums readily developed by available
equipment
AMMONIA VAPOR PRESSURE
TABLE 2
