Measuring temperature with the thermistor, History – PASCO TD-8551A MECHANICAL EQUIVALENT OF HEAT User Manual
Page 6
Mechanical Equivalent of Heat
012-04331E
2
soldered to the copper slip rings (see Figure 3) on the side of
the cylinder. The brushes provide an electrical connection
between the slip rings and the banana plug connectors. By
plugging an ohmmeter into these connectors, the resistance
of the thermistor, and therefore it's temperature, can be
monitored, even when the cylinder is turning.
Although the temperature dependence of the thermistor is
accurate and reliable, it is not linear. You will therefore
need to use the table of Temperature versus Resistance that
is affixed to the base of the Mechanical Equivalent of Heat
apparatus to convert your resistance measurements into
temperature readings. A more complete version of this
table, covering a greater temperature range, is given at the
end of this manual.
Measuring Temperature with the Thermistor
It may not seem strange to us today that there is a thing
called energy that is conserved in all physical interactions.
Energy is a concept we have all grown up with. A hundred
and fifty years ago it was not so evident that there should be
an intimate, quantitative relationship between such appar-
ently unrelated phenomena as motion and heat. The
discovery that heat and motion can be seen as different
forms of the same thing—namely energy—was the first and
biggest step toward understanding the concept of energy
and its conservation.
Count Rumford of Bavaria, in 1798, was the first to realize
that work and heat were related phenomena. At that time, it
was commonly believed that heat resulted from the flow of
a massless fluid-like substance called caloric. It was
believed that this substance resided in objects, and that
when they were cut, ground, or otherwise divided into
smaller pieces, the pieces could not hold as much caloric as
the original object. The resulting release of caloric was
what we experience as heat.
While boring cannon for the Bavarian government,
Rumford noticed that heat was produced even when the
boring equipment had become so dulled from use that it was
no longer boring into the iron. The heat therefore was not
dependent on the breaking up of the metal into smaller
pieces. In fact, this meant that a limitless amount of heat
could be produced from the iron and boring equipment, an
idea that was inconsistent with the belief that heat was the
result of the release of a substance that resided in the
material. Rumford realized that a connection existed
between the motion of the bore and the heat. He even took
his reasoning a step further, stating his belief that only if
heat were a form of motion would it demonstrate the
properties he had observed.
It was not until the experiments of Joule in 1850, however,
that Rumford's ideas about the nature of heat gained popular
acceptance. Joule performed a variety of experiments in
which he converted a carefully measured quantity of work,
through friction, into an equally carefully measured quantity
of heat. For example, in one experiment Joule used falling
masses to propel a paddle wheel in a thermally insulated,
water-filled container. Measurements of the distance
through which the masses fell and the temperature change
of the water allowed Joule to determine the work performed
and the heat produced. With many such experiments, Joule
demonstrated that the ratio between work performed and
heat produced was constant. In modern units, Joule's results
are stated by the expression:
1 calorie = 4.186 Joule.
Joule's results were within 1% of the value accepted today.
(The calorie is now defined as equal to 4.184 Joule.)
It was this series of experiments that led Joule, along with
several others, to the more general theory that energy is
conserved in all physical processes.
History
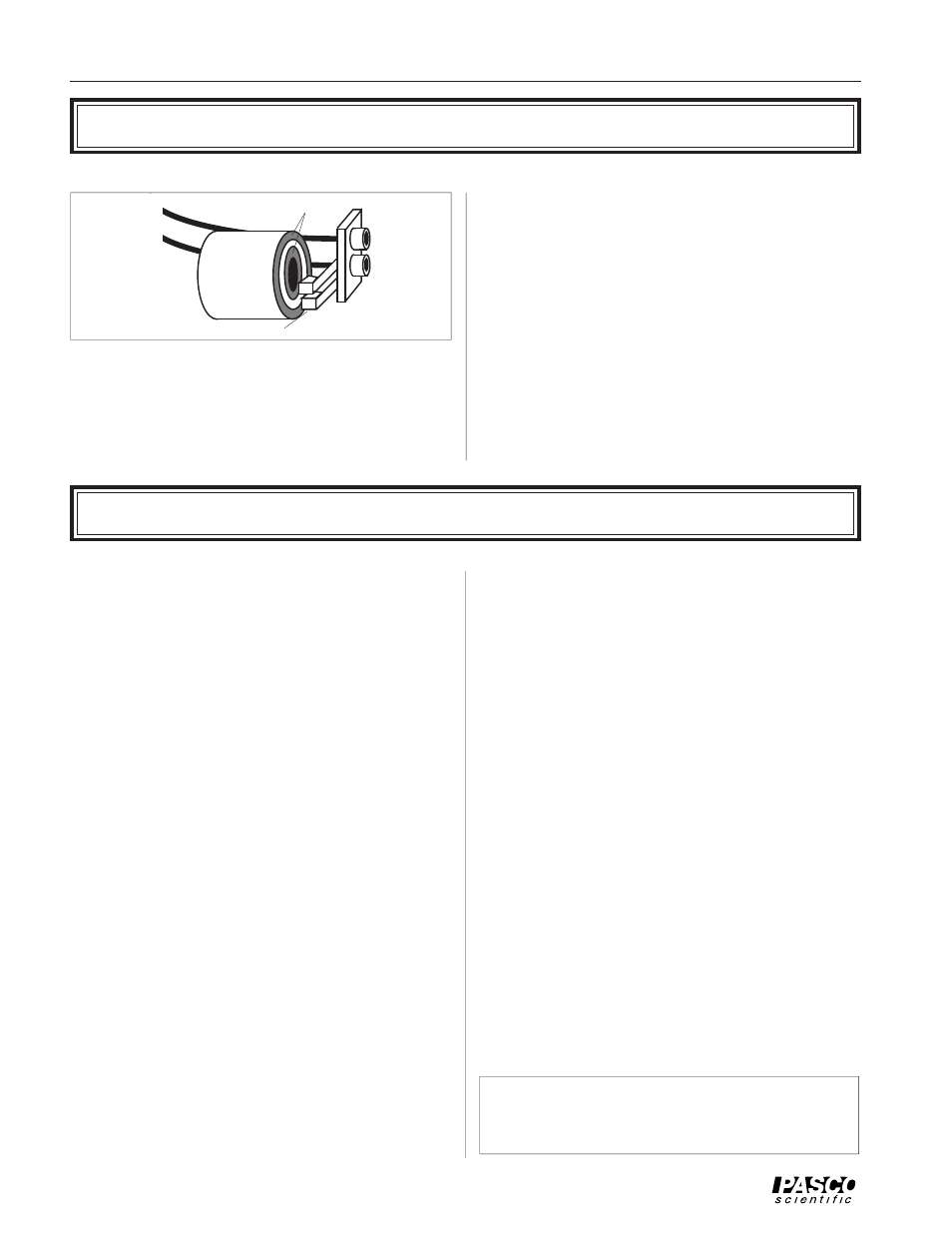
To measure the temperature of the aluminum cylinder, a
thermistor is embedded inside. A thermistor is a tempera-
ture dependent resistor. If the resistance of the thermistor is
known, its temperature can be very accurately and reliably
determined. The leads of the thermistor in the cylinder are
Figure 3 Measuring the Cylinder Temperature
Slip Rings
Banana
Jacks
Brushes
To
Ohmmeter
➤ NOTE: See the short biography at the end of
this manual for more information on the life of
Benjamin Thompson—Count Rumford, of Bavaria.
