UEi Test Instruments C127OILKIT User Manual
Page 13
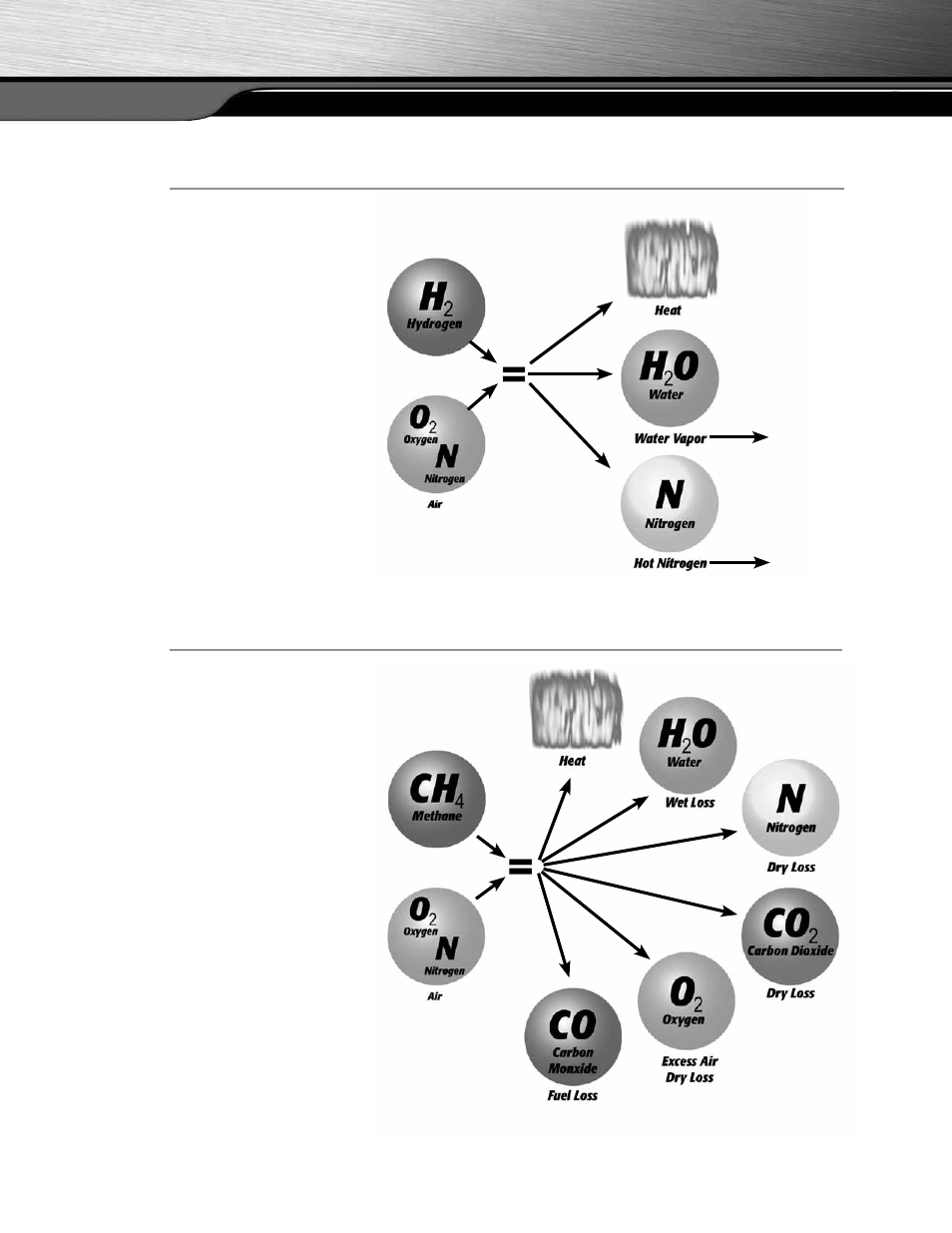
N
ear
i
deal
C
ombuStioN
When we burn pure
hydrogen in the air. Our atmo-
sphere is 20.9% oxygen with the
remaining 79.1% nitrogen.
This is nearly as desirable as the
example for ideal combustion
with the only added loss being
the heat that is carried away from
your target with the nitrogen.
Because nitrogen isn’t part of the
combustion process, it enters the
combustion chamber at the inlet
temperature and leaves with some
of the heat created by the com-
bustion. If this isn’t recovered
at the heat exchanger it is lost up
the flue.
The main problem with this
example is again the availability
and cost of pure hydrogen.
b
eSt
o
f
t
he
r
eal
w
orld
Natural gas is a readily
available fuel, and our atmosphere
contains sufficient oxygen. When
this is used as a fuel we get the
reaction; shown in figure 3.
Now the other added byproducts
are CO2 and hot nitrogen com-
pared to the Ideal World situation.
In addition to this we have added
the byproduct Excess Air.
Excess Air is exactly what the name
implies, air that is in excess of what
is needed to burn all of the fuel.
The reason for this is more related
to the ability to mix all of the fuel
and O2 for complete combustion.
Without some amount of excess air
not all of the fuel would burn com-
pletely, and this leads to the forma-
tion of CO instead of CO2.
Other fuels all contain the basic
ingredients for combustion, but also
may include other components such
as sulfur, fuel bound nitrogen, soot
and ash and water. These either
react with the oxygen to form other
pollutants or contribute to
additional losses.
Wet Loss
Dry Loss
figure 2
figure 3
Carbon Monoxide
is formed from incomplete combustion (partial oxidation of carbon in
the fuel). Typical causes are incomplete mixing of fuel and air, low combustion temperatures,
or not enough excess air.
12
13
