Hanna Instruments HI 83740 User Manual
Page 6
6
PRINCIPLE OF OPERATION
Absorption of Light is a typical phenomenon of interaction between electromagnetic radiation and
matter. When a light beam crosses a substance, some of the radiation may be absorbed by atoms,
molecules or crystal lattices.
If pure absorption occurs, the fraction of light absorbed depends both on the optical path length
through the matter and on the
physical
-chemical characteristics of the substance according to the
Lambert-Beer Law:
-log
I
/
I
o
=
ε
λ
c d
or
A
=
ε
λ
c d
Where:
-log
I
/
I
o
= Absorbance (A)
I
o
= intensity of incident light beam
I
= intensity of light beam after absorption
ε
λ
= molar extinction coefficient at wavelength
λ
c
= molar concentration of the substance
d
= optical path through the substance
Therefore, the concentration "c" can be calculated from the absorbance of the substance as the
other factors are known.
Photometric chemical analysis is based on the possibility to develop an absorbing compound from
a specific chemical reaction between sample and reagents. Given that the absorption of a
compound strictly depends on the wavelength of the incident light beam, a narrow spectral
bandwidth should be selected as well as a proper central wavelength to optimize measurements.
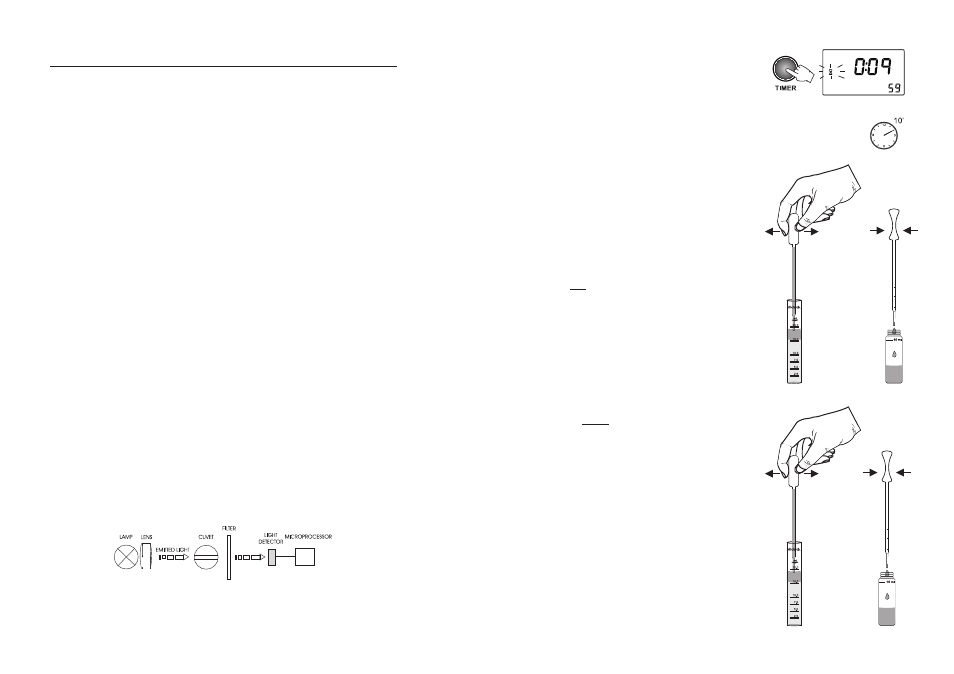
The optical system of Hanna's HI 83000 series colorimeters is based on special subminiature
tungsten lamps and narrow-band interference filters to guarantee both high performance and
reliable results.
Block diagram (optical layout)
15
• Press TIMER and the instrument will show the
countdown or, alternatively, wait for 10 minutes,
leaving the vials capped and undisturbed.
During this period the color of the upper layer (organic
phase) in vial #2 will turn purple if copper is
present.
After 10 minutes the instrument gives an acoustic
signal to alert the user that the countdown has finished.
• Remove the cap of vial #1. Use the 3 mL plastic
pipette to transfer the upper layer (organic phase) into
a cuvet. Ensure that at least 1/3 of the cuvet is
filled with organic solvent (see page 11).
If some wine is transferred too, this does not interferes
with the measurement.
Cap the cuvet. This is the zero (#1).
Note:
The upper layer is an emulsion of small wine drops
dispersed in the organic phase. Please note that an
emulsion is an unstable equilibrium that may separate
even after few minutes. It is therefore important to
measure both the zero and the sample immediately
after the countdown has finished.
In case the emulsion separates before measurements
can be made, we recommend to leave the vials standing
for at least 4 hours, allowing complete separation of
the emulsion and obtaining two clear solutions in the
cuvets. Since the developed color is very stable, the
cuvets may be left standing overnight to be read also
next morning.
• Remove the cap of vial #2. Use the 3 mL plastic
pipette to transfer the upper layer (organic phase) into
another empty cuvet (see page 11).
If some wine is transferred too, this does not interferes
with the measurement.
Cap the cuvet. This is the reacted sample (#2).
#2
(Sample)
#1
(Zero)
