Method guidelines, I. principles of oligonucleotide separations, Ii. sample preparation – Waters Oligonucleotide Separation Technology ACQUITY UPLC C18 Column User Manual
Page 2
2
[ method guidelines ]
I. PrInCIPLes oF oLIgonUCLeotIde sePArAtIons
Separations of detritylated synthetic oligonucleotides on ACQUITY
UPLC
®
OST C
18
columns are based on ion-pair, reversed-phase chro-
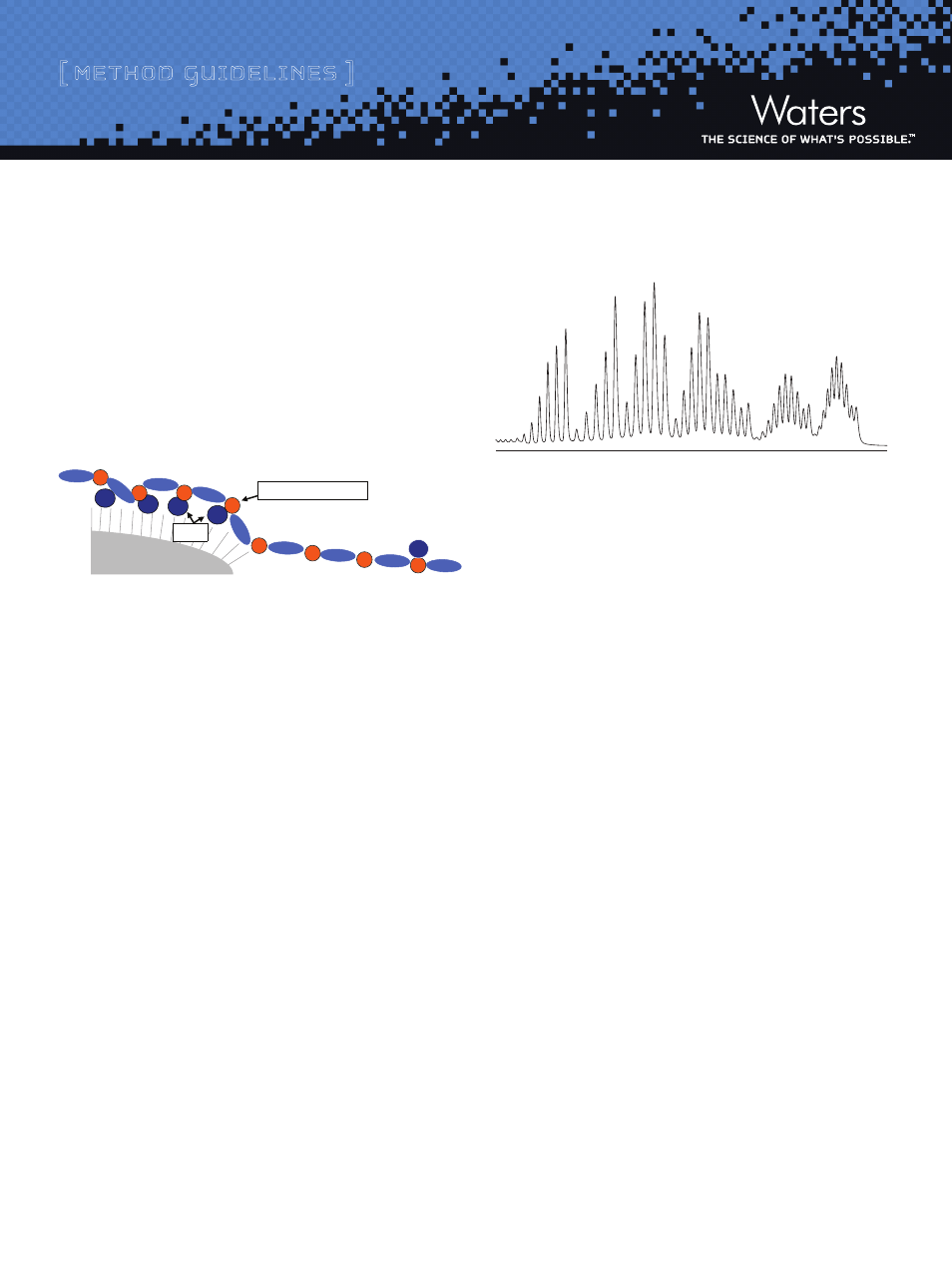
matographic principles (IP-RP-LC). As shown in Figure 1, the ion-pair-
ing additive in the mobile phase is adsorbed on a hydrophobic sorbent
and provides for charge-to-charge interactions with negative charges
contained on the oligonucleotide backbone (i.e., phosphate groups).
Figure 1: Proposed Mechanism of IP-RP-LC for Synthetic Oligonucleotide
Separations
As a result, an efficient charge-based (length-based) oligonucleotide
separation is achieved (Figure 2). Gradient elution using an aceto-
nitrile or methanol eluent displaces both ion-pairing agent and the
oligonucleotides from the sorbent surface.
Separation selectivity and resolution decreases with increasing
oligonucleotide length (Figure 2) making the separation of long
oligonucleotides challenging. Modified oligonucleotides such as phos-
phorothioates and 2-O alkyl modified species are also more difficult
to analyze. Special mobile phase may be required (see Section III,
Recommended Mobile Phases).
Figure 2: Separation of a 15 - 60mer Deoxythymidine Ladder on
ACQUITY UPLC
®
OST C
18
Column
UPLC
®
System:
Waters ACQUITY UPLC
®
System with installed 425 µL mixer
PDA Detector with standard UV cell
Sample Injected:
Approximately 100 pmoles of a detritylated 15 – 60mer
oligonucleotide ladder diluted in 0.1 M TEAA
Column:
Waters ACQUITY UPLC
®
OST C
18
column, 1.7 µm (2.1 x 50 mm)
Mobile Phases:
A: 0.1 M TEAA,
B: Acetonitrile / 0.1M TEAA, 20/80, v/v
Flow rate:
0.2 mL/min
Column Temp.:
60 ˚C
Gradient delay:
0.45 mL
Gradient:
50 to 65% B in 20 minutes (10-13% acetonitrile,
0.15% acetonitrile per min)
Detection:
260 nm, 20 scans per second
II. sAMPLe PrePArAtIon
Follow these steps to prepare your sample:
1. Dissolve the detritylated synthetic oligonucleotide sample in
Mobile Phase A (e.g., 0.1 M TEAA). For example, a 0.05 - 0.2
µmole scale synthesis can be prepared in 0.1 mL of 0.1 M TEAA.
Proportionately larger or smaller volumes of 0.1 M TEAA are
required when dissolving samples from different scale syntheses.
Due to the nature of gradient separations, relatively large volumes
of sample (in low organic strength eluent) can be injected and
concentrated onto the head of the column before beginning the
gradient elution program.
2. Samples must be completely in solution and free of particulates.
Remove all particles from the sample (Controlled Pore Glass
Synthesis Support, etc.), which may block the inlet column frit,
increase the operating pressure, and shorten the column life time.
Sample contamination with high concentration of salts and/or
detergents may also interfere with analysis.
+
+
+
+
-
-
-
-
-
-
-
-
+
T EA
ACQUITY UPLC
®
OST C
18
chain
PO group on Oligo chain
2
minutes
16
60
20
25
30
35
40
50
